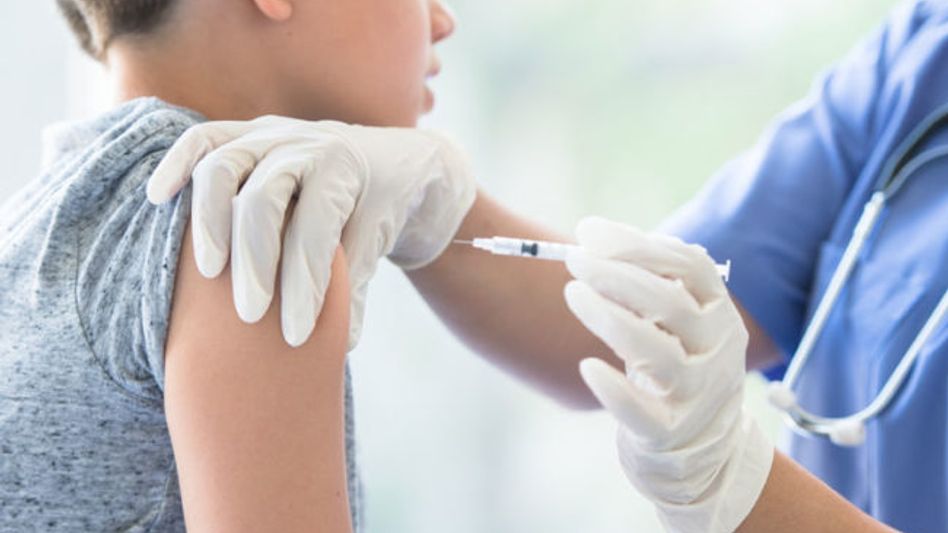
Centre denies permission to SII to conduct Covavax trials on children
The central government on Thursday recommended against allowing Serum Institute of India to conduct phase 2 and 3 clinical trials of Covavax COVID-19 vaccine for the age group 2-17 years.
 iNDIA
iNDIANEW DELHI: The central government on Thursday recommended against allowing Serum Institute of India to conduct phase 2 and 3 clinical trials of Covavax COVID-19 vaccine for the age group 2-17 years.
The government instructed SII to complete the trials on adults first.
As children are more likely to be affected by the third wave of COVID-19, vaccines for them has become the need of the hour.
Also Read: Breaking: NIA court discharges Akhil Gogoi from remaining Chandmari case
Reportedly the centre recommended that "the Pune-based company should submit the safety and immunogenicity data (of Covovax) from the ongoing clinical trial in adults for considering the conduct of a clinical trial in children," as stated in a report.
SII had sought permission from Drug Controller General of India to test the vaccine on children.
Earlier, the Centre said that Novavax COVID vaccine efficacy data in a large trial are promising.
Also the clinical trials are being conducted and are in an advanced stage of completion in India.
Copyright©2026 Living Media India Limited. For reprint rights: Syndications Today









